Korean-Developed Humanized Mouse Model Exported to UK for Antibody Therapy Research
Translated from Korean, summarized and contextualized by DistantNews.
TLDR
- GEMCRO, a South Korean company specializing in genetically modified mice, has exported its hiFcRn™ humanized mouse model to a UK-based antibody therapy developer.
- This marks the first instance of a domestic preclinical animal model being directly utilized in overseas new drug development research.
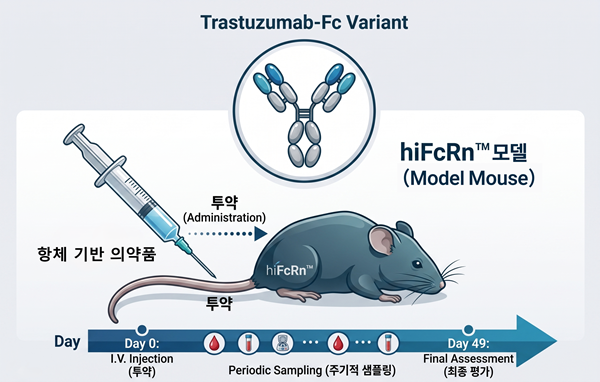
- The hiFcRn™ model precisely mimics the human antibody recycling system, allowing for more accurate prediction of antibody-based drug efficacy and pharmacokinetics.
Dong-A Ilbo highlights a significant achievement for South Korea's biotechnology sector: GEMCRO's successful export of its proprietary hiFcRn™ humanized mouse model to the United Kingdom. This event is particularly noteworthy as it represents the first time a preclinical animal model developed in Korea has been directly integrated into foreign new drug development research. The hiFcRn™ model, designed to accurately simulate the human FcRn (neonatal Fc receptor) system, is crucial for evaluating the efficacy and pharmacokinetic properties of antibody-based therapeutics, offering a more clinically relevant prediction than previous models. GEMCRO's expertise in gene editing technologies like CRISPR/Cas9 and its robust supply chain for frozen sperm and embryos underscore its advanced capabilities. This export not only validates GEMCRO's technical prowess but also positions South Korea as a key player in the global preclinical research market. The company's ambition to expand into European and North American markets following this success signals a promising future for Korean biotech innovation on the international stage.
This export is significant in that a preclinical animal model developed in Korea has been directly incorporated into the global new drug development pipeline.
Originally published by Dong-A Ilbo in Korean. Translated, summarized, and contextualized by our editorial team with added local perspective. Read our editorial standards.