Rare skin condition patients gain access to new treatment
Summarized and contextualized by DistantNews.
TLDR
- Ireland has approved a new gel treatment, Filsuvez, for Epidermolysis Bullosa (EB), a painful skin condition.
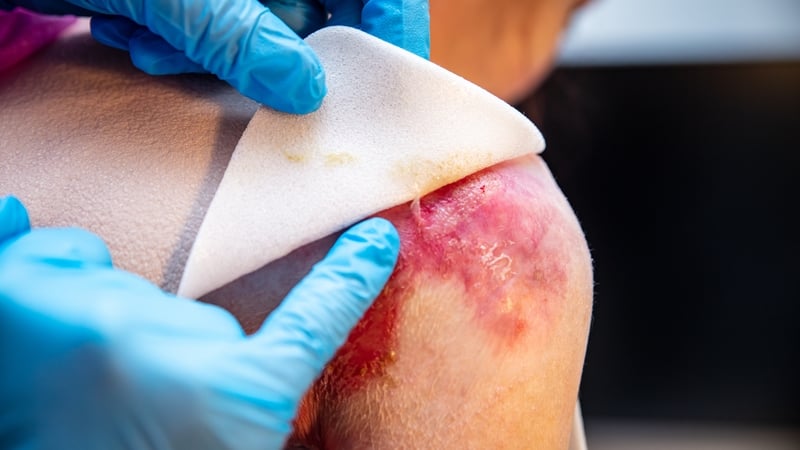
- The treatment promotes wound healing and can ease pain for EB patients, also known as "butterfly skin."
- The HSE will cover most of the cost, marking a significant step for the 300 people in Ireland affected by EB.
This RTÉ News report heralds a significant breakthrough for individuals in Ireland living with Epidermolysis Bullosa (EB), a rare and debilitating skin condition. The approval and availability of Filsuvez, the first authorized treatment for EB, is framed as a "transformative moment" by Debra, the national charity supporting patients.
This has been a long journey and marks a transformative moment for people living with this rare skin condition.
The article emphasizes the profound impact this new gel treatment will have on the lives of approximately 300 people in Ireland battling EB, often referred to as "butterfly skin" due to the extreme fragility of their skin. By promoting wound healing and reducing pain associated with bandage changes, Filsuvez offers much-needed relief and improved quality of life. The HSE's commitment to covering the cost underscores the healthcare system's recognition of the urgent unmet need within this community.
This decision represents hope, dignity and meaningful progress for EB patients and their families.
From an Irish perspective, this news is particularly poignant. Debra chief executive Jimmy Fearon's statement that "Ireland is in the lowest quartile in Europe for introducing new drugs to the market" highlights a systemic issue that this approval helps to address. The lengthy average timeline for drug reimbursement in Ireland compared to the rest of Europe is a source of frustration for patient groups. Therefore, the swift action on Filsuvez is not just about a new treatment; it's about a hopeful sign that the system may be improving, offering "hope, dignity and meaningful progress" to patients and their families.
For too long, people living with EB have had no approved treatment options.
This story is uniquely interesting from an Irish viewpoint because it directly confronts the challenges faced by patients in accessing cutting-edge medical treatments. While Western media might report on the drug's efficacy, the Irish coverage, as seen here, delves into the specific context of Ireland's drug approval process and the advocacy efforts that led to this positive outcome. It celebrates a victory for patient advocacy and a step towards greater equity in healthcare access within the country.
Ireland is in the lowest quartile in Europe for introducing new drugs to the market.
Originally published by RTÉ News. Summarized and contextualized by our editorial team with added local perspective. Read our editorial standards.